SUGAR (Ligand Exchange Chromatography)
Polymer-based columns for the analysis of saccharides, sweeteners, sugar alcohols and moisterizers
Product overview
SUGAR SC1011
SUGAR SC1211
Columns with calcium (Ca2+) counter ions
SUGAR SC1011
-
For saccharide analysis using ligand exchange mode
-
Only water (or Ca-salt solution) is required for the analysis of neutral sugars
-
Fulfills USP L19 and L22 requirements
SUGAR SC1211
-
Combination of ligand exchange and HILIC modes
-
Suitable for separating sugar alcohols
-
Fulfills USP L19 and L22 requirements


SUGAR KS-800 series
Columns with sodium (Na+) counter ions
-
Separates saccharides by combination of ligand exchange and size exclusion modes
-
Only water is required for the analysis of neutral sugars
-
Suitable for separation of polysaccharides by size exclusion mode
-
Can be used in combination (2-3 column in row with same or different exclusion limit)
-
Fulfill USP L22 and L58 requirements
Column Selection Saccharides

Theory about Ligand Exchange Chromatography
HPLC-based analyses of saccharides and sugar alcohols are performed in various modes, such as HILIC, ligand exchange, size exclusion, and ion exchange.
In the SUGAR series columns, a hard styrene-divinylbenzene copolymer designed for saccharide analysis is used as the base material, and a strong cation exchange resin incorporating functional sulfo-groups coupled with metal counter ions is used as the packing. Columns of the SUGAR series are available in various types according to counter ion, pore size, and pore structure, showing differential selectivity for different saccharides.
The series are suitable for saccharide separation and analysis of foods, areas of biochemistry, and natural substances. Other items include the Asahipak GS-220 HQ polymeric packed columns for size exclusion mode to analyze water-soluble, low molecular weight dietary fibers, and the OHpak SB-800 HQ polymeric packed columns for size exclusion mode to analyze polysaccharides, as well as the SUGAR KS-800 columns.
A strong feature of the Shodex SUGAR series columns resides in the mechanism of separation in the ligand exchange mode. Ligand exchange refers to a mode of separation based on the interaction (ligand exchange potential) between hydroxyl groups and metal ions to form a complex.
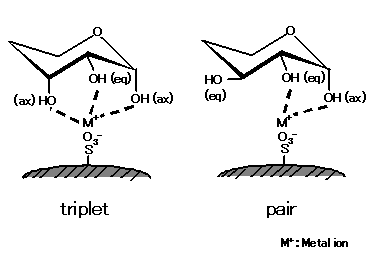
Saccharides exhibits an energy-stable chair conformation in 5-membered ring (furanose) or 6-membered ring (pyranose) forms. Since hydroxyl group on each carbon can take either equatorial or axial position, even two saccharides having the same molecular structures may have different three dimensional configurations. Ligand exchange mode separates saccharides using this configuration difference of the complex formed between saccharides' hydroxyl groups and metal ions.
As shown in the left figure, saccharides having a larger number of ax-eq-ax configuration (triplet) units form stronger complexes with metal ions. Meanwhile, as right figure shows, saccharides lacking such a triplet structure form complexes with ax-eq (pair) hydroxyl group. As the unit number of this pair structure increases, saccharides become more potent forming complexes with metal ions. The complex formation capacity also differs depending on modified metal ions.
The complex formation potential also differs depending on the kind of metal ion.
Ag+ < Li+ < Na+ < Zn2+ < Ca2+ < Ba2+ < Pb2+
When using a column of the SUGAR series, analysis should be performed at increased column temperatures to prevent anomer separation. Under strongly alkaline conditions, saccharides are likely to isomerize with the fear of decomposition of polysaccharides.